Lesson 3
3.2.4 Macromolecules
• Describe the giant covalent structures of graphite and diamond
• Relate their structures to their uses, e.g. graphite as a lubricant and a conductor, and
diamond in cutting tools
• Describe the similarity in properties between diamond and silicon(IV) oxide, related to their
structures
3.2.5 Metallic bonding
• Describe metallic bonding as a lattice of positive ions in a ‘sea of electrons’ and use
this to describe the electrical conductivity and malleability of metals
• Describe qualitatively the pressure and temperature of a gas in terms of the motion
of its particles
LESSONPART 1
3.2.4 Macromolecules
• Describe the giant covalent structures of graphite and diamond
- Diamond and graphite are allotropes of carbon which have giant covalent structures.
- These classes of substance contain a lot of non-metal atoms, each joined to adjacent atoms by covalent bonds forming a giant lattice structure.
- Giant covalent structures have high melting and boiling points as they have many strong covalent bonds that need to be broken down.
- Large amounts of heat energy are needed to overcome these forces and break down bonds.
diamond in cutting tools
Diamond
• Describe the macromolecular structure of silicon(IV) oxide (silicon dioxide)- Each carbon atom bonds with four other carbons, forming a tetrahedron.
- All the covalent bonds are identical and strong with no weak intermolecular forces.
- Diamond thus:
- Does not conduct electricity.
- Has a very high melting point.
- Is extremely hard and dense (3.51 g/cm3).
- Diamond is used in jewellery and as cutting tools.
- The cutting edges of discs used to cut bricks and concrete are tipped with diamonds.
- Heavy-duty drill bits and tooling equipment are also diamond tipped.
- Each carbon atom is bonded to three others forming layers of hexagonal shaped forms, leaving one free electron per carbon atom.
- These free electrons exist in between the layers and are free to move and carry charge, hence graphite can conduct electricity.
- The covalent bonds within the layers are very strong but the layers are connected to each other by weak intermolecular forces only, hence the layers can slide over each other making graphite slippery and smooth.
- Graphite thus:
- Conducts electricity.
- Has a very high melting point.
- Is soft and slippery, less dense than diamond (2.25 g/cm3).
- Graphite is used in pencils and as an industrial lubricant, in engines and in locks.
- It is also used to make non-reactive electrodes for electrolysis.
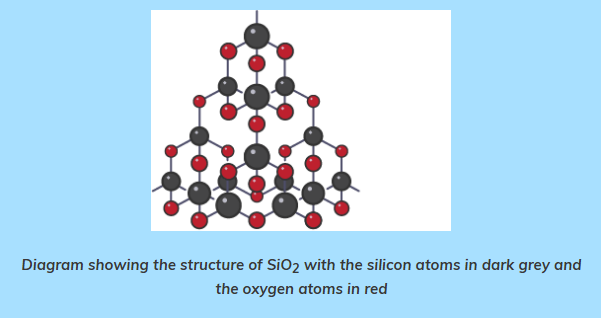
- SiO2 is a macromolecular compound which occurs naturally as sand and quartz.
- Each oxygen atom forms covalent bonds with 2 silicon atoms and each silicon atom in turn forms covalent bonds with 4 oxygen atoms.
- A tetrahedron is formed with one silicon atom and four oxygen atoms, similar as in diamond.
structures
- SiO2 has lots of very strong covalent bonds and no intermolecular forces so it has similar properties as diamond.
- It is very hard, has a very high boiling point, is insoluble in water and does not conduct electricity.
- SiO2 is cheap since it is available naturally and is used to make sandpaper and to line the inside of furnaces.
• Describe metallic bonding as a lattice of positive ions in a ‘sea of electrons’ and use
this to describe the electrical conductivity and malleability of metals
- Metal atoms are held together strongly by metallic bonding.
- Within the metal lattice, the atoms lose their valence electrons and become positively charged.
- The valence electrons no longer belong to any metal atom and are said to be delocalised.
- They move freely between the positive metal ions like a sea of electrons.
- Metallic bonds are strong and are a result of the attraction between the positive metal ions and the negatively charged delocalised electrons.
The link between metallic bonding and the properties of metals:
---------------------------------------------------------------------------------------------- Metals have high melting and boiling points:
- There are many strong metallic bonds in giant metallic structures.
- A lot of heat energy is needed to overcome forces and break these bonds.
- Metals conduct electricity:
- There are free electrons available to move and carry charge.
- Electrons entering one end of the metal cause a delocalised electron to displace itself from the other end.
- Hence electrons can flow so electricity is conducted.
- Metals are malleable and ductile:
- Layers of positive ions can slide over one another and take up different positions.
- Metallic bonding is not disrupted as the valence electrons do not belong to any particular metal atom so the delocalised electrons will move with them.
- Metallic bonds are thus not broken and as a result metals are strong but flexible.
- They can be hammered and bent into different shapes without breaking.
LESSON PART 2
1 The particulate nature of matter
1.1 The particulate nature of matter
• State the distinguishing properties of solids, liquids and gases
• Describe the structure of solids, liquids and gases in terms of particle separation,
arrangement and types of motion
1.1 The particulate nature of matter
• State the distinguishing properties of solids, liquids and gases
- Solids, liquids and gases have different physical properties.
- The difference in these properties comes from differences in how the particles are arranged in each state.
arrangement and types of motion
Solids
- Strong forces of attraction between particles, particles are packed very closely together in a fixed and regular pattern.
- Atoms vibrate in position but can’t change position or move.
- Solids have a fixed volume, shape and high density.
- Weaker attractive forces in liquids than in solids, particles are close together in an irregular, unfixed pattern.
- Particles can move and slide past each other which is why liquids adopt the shape of the container they’re in and also why they are able to flow.
- Liquids have a fixed volume but not a fixed shape and have a moderate to high density.
- No intermolecular forces, particles are in random movement and so there is no defined pattern.
- Particles are far apart and move quickly (around 500 m/s) in all directions, they collide with each other and with the sides of the container (this is how pressure is created inside a can of gas).
- No fixed volume, since there is a lot of space between the particles, gases can be compressed into a much smaller volume. Gases have low density.
• Describe changes of state in terms of melting, boiling, evaporation, freezing,
condensation and sublimation
condensation and sublimation
- The presence of impurities in a pure substance can change its melting point and boiling point (m.p. & b.p.).
- Different pure substances can be identified by analysis of the value of their m.p. or b.p. since this is a physical property which is unique to each substance.
Melting
- Melting is when a solid changes into a liquid.
- Requires heat energy which transforms into kinetic energy, allowing the particles to move.
- Occurs at a specific temperature known as the melting point (m.p.) which is unique to each pure solid.
Boiling
- Boiling is when a liquid changes into a gas.
- Requires heat which causes bubbles of gas to form below the surface of a liquid, allowing for liquid particles to escape from the surface and within the liquid.
- Occurs at a specific temperature known as the boiling point (b.p.) which is unique to each pure liquid.
Freezing
- Freezing is when a liquid changes into a solid.
- This is the reverse of melting and occurs at exactly the same temperature as melting, hence the melting point and freezing point of a pure substance are the same. Water for example freezes and melts at 0ºC.
- Requires a significant decrease in temperature (or loss of thermal energy) and occurs at a specific temperature which is unique for each pure substance.
Evaporation
- When a liquid changes into a gas. Evaporation occurs only at the surface of liquids where high energy particles can escape from the liquid’s surface at low temperatures, below the b.p. of the liquid.
- The larger the surface area and the warmer the liquid/surface, the more quickly a liquid can evaporate
- No heat is required and evaporation occurs over a range of temperatures.
Condensation
- When a gas changes into a liquid, usually on cooling. When a gas is cooled its particles lose energy and when they bump into each other, they lack energy to bounce away again, instead grouping together to form a liquid.
- No energy is required for condensation to occur and it takes place over a range of temperatures.
Sublimation
- When a solid changes directly into a gas.
- This happens to only a few solids such as iodine or solid carbon dioxide.
- The reverse reaction also happens and is also called sublimation (sometimes called deposition or desublimation).
- Sublimation occurs at a specific temperature which is unique for a pure substance.
- Questions on the particle theory of matter show interconversion of states with a reversible arrow: ⇌, which means that the process can go forwards and backwards.
- Read the question carefully and pick the direction of the change in state that the question refers to.
of its particles
Gaseous Particles
- Gaseous particles are in constant and random motion.
- An increase in temperature increases the kinetic energy of each particle, as the thermal energy is transformed to kinetic energy, so they move faster.
- Decreasing the temperature has the opposite effect.
- The pressure that a gas creates inside a closed container is produced by the gaseous particles hitting the inside walls of the container. As the temperature increases, the particles in the gas move faster, impacting the container’s walls more frequently.
- Therefore an increase in temperature causes an increase in pressure.
• Explain changes of state in terms of the kinetic theory
- When substances are heated, the particles absorb thermal energy which is converted into kinetic energy. This is the basis of the kinetic theory of matter.
- Heating a solid causes its particles to vibrate more and as the temperature increases, they vibrate so much that the solid expands until the structure breaks and the solid melts.
- On further heating, the now liquid substance expands more and some particles at the surface gain sufficient energy to overcome the intermolecular forces and evaporate.
- When the b.p. temperature is reached, all the particles gain enough energy to escape and the liquids boils.
- These changes in state can be shown on a graph which is called a heating curve.
- Cooling down a gas has the reverse effect and this would be called a cooling curve.
- These curves are used to show how changes in temperature affect changes of state.
-
- While changing state, the temperature of the substance remains the same as the heat energy is rapidly converted into kinetic energy.
- This is called latent heat and corresponds to the horizontal sections of a heating / cooling curve.


















No comments:
Post a Comment